
Photo via Pexels
A team led by researchers from the U.S. Department of Energy's Argonne National Laboratory and the University of Toronto has developed a novel electrocatalyst that directly converts carbon dioxide into ethanol. This copper-based catalyst achieved a remarkable 60% selectivity for ethanol production, minimizing unwanted byproducts. The innovative methodology involves a unique copper nanostructure that efficiently facilitates the multi-electron transfer steps required for CO2 reduction to ethanol. The surprising implication is that a common greenhouse gas could become a direct feedstock for a clean-burning liquid fuel, rather than merely being captured or stored. This work was published in *Nature Energy*.
Why It’s Fascinating
This discovery is highly significant because previous CO2 conversion efforts often yielded less valuable single-carbon products like carbon monoxide or methane, or required multiple energy-intensive steps. It overturns the prior understanding that efficient, direct electrocatalytic conversion of CO2 to complex liquid fuels like ethanol was prohibitively difficult at scale. Within 5-10 years, this technology could be integrated into industrial processes to convert emissions from power plants or factories into usable fuel, offering a tangible path to a circular carbon economy. Think of it as a chemical 'photosynthesis' for industrial waste, mimicking nature's ability to turn CO2 into useful compounds. Energy companies, industrial polluters, and policymakers pushing for decarbonization stand to benefit most. What if every major CO2 emitter could become an ethanol producer? This directly competes with other CO2 utilization strategies that produce less energy-dense or less readily usable products.
Related
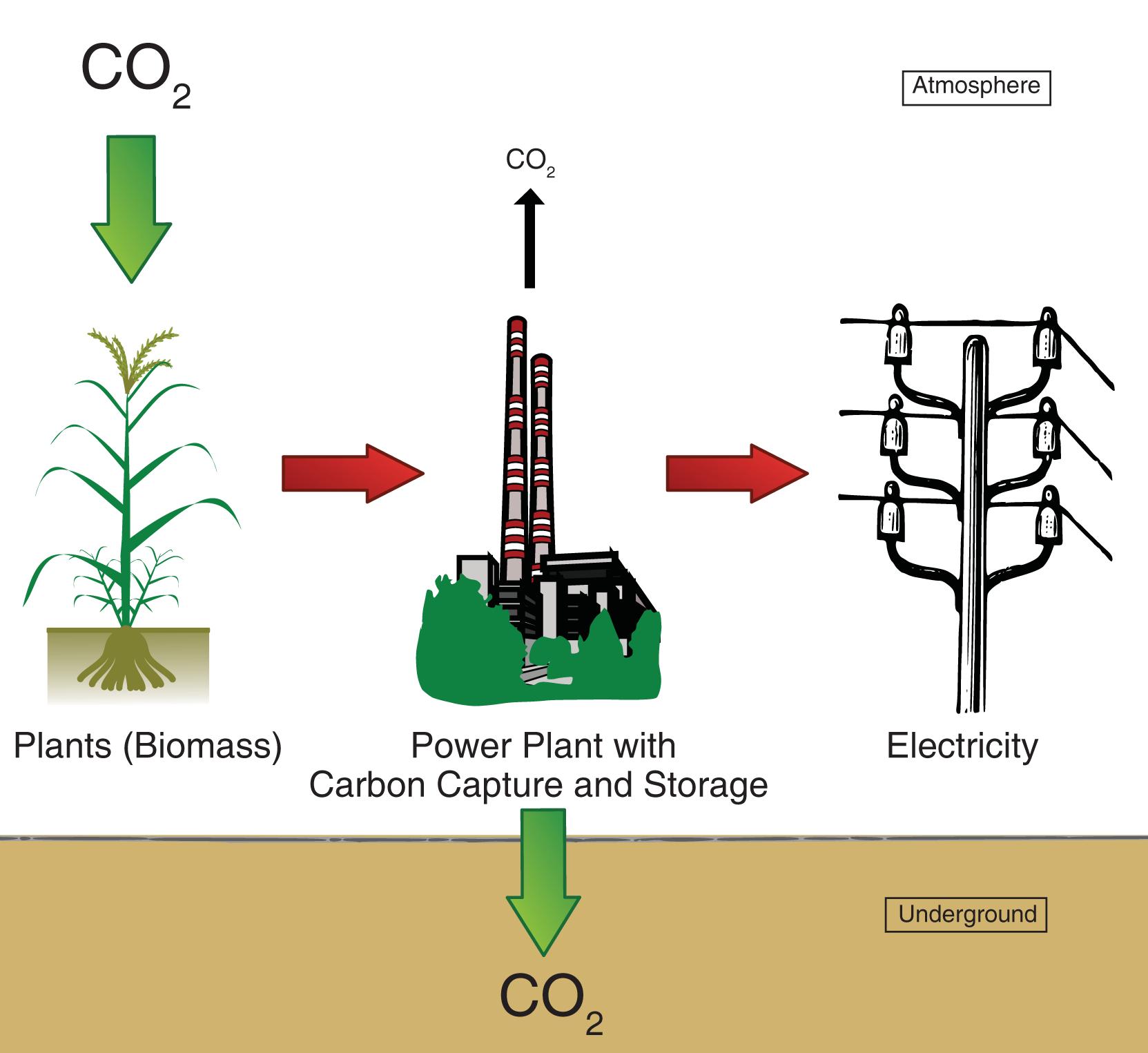
Bioenergy Carbon Capture and Storage (BECCS)
Bioenergy with Carbon Capture and Storage (BECCS) combines the generation of energy from biomass (e.g., agricultural waste, dedicated energy crops) with…

Omnivore
Omnivore is a free and open-source read-it-later application developed by a community-driven project, designed for saving articles, newsletters, and web pages…

Zotero
Zotero, a free and open-source project developed by the Roy Rosenzweig Center for History and New Media at George Mason University, is a powerful reference…

Goal Zero Nomad 20 Solar Panel
The Goal Zero Nomad 20 Solar Panel is a highly portable and efficient power source. This compact 20-watt monocrystalline solar panel efficiently converts…
More from Discoveries
View all →
AI Deciphers Lost Language of Ancient Civilization
Read →
Ancient Microbes Revived From Salt Crystals
Read →
Fungi 'Talk' Via Electrical Signals
Read →
Black Hole Jets Baffle Physicists by Bending Light
Read →
Ancient DNA Rewrites History of Nomadic Empires
Read →
Quantum Entanglement Stabilizes Fragile Molecules
Read →
AI Deciphers Lost Language of Ancient Civilization
Read →
Ancient Microbes Revived From Salt Crystals
Read →
Fungi 'Talk' Via Electrical Signals
Read →
Black Hole Jets Baffle Physicists by Bending Light
Read →
Ancient DNA Rewrites History of Nomadic Empires
Read →
Quantum Entanglement Stabilizes Fragile Molecules
Read →Enjoyed this? Get five picks like this every morning.
Free daily newsletter — zero spam, unsubscribe anytime.